Water quality is usually described by different indicators such as temperature, dissolved oxygen, pH, total dissolved solids, conductivity, suspended sediment, nutrients, bacteria, metals, hydrocarbons and industrial chemicals.
Water quality is one of the most important factors in aquatic ecosystems, ensuring that water is safe for human use. Actions taken on land have a major impact on what happens in water-based ecosystems, which is why monitoring water quality levels is so important.
Assessing water quality usually involves comparing measured chemical concentrations with natural concentrations, background or baseline concentrations, and guidelines established to protect human health or ecological communities.
7 Main water quality indicators
Temperature and dissolved oxygen (DO)
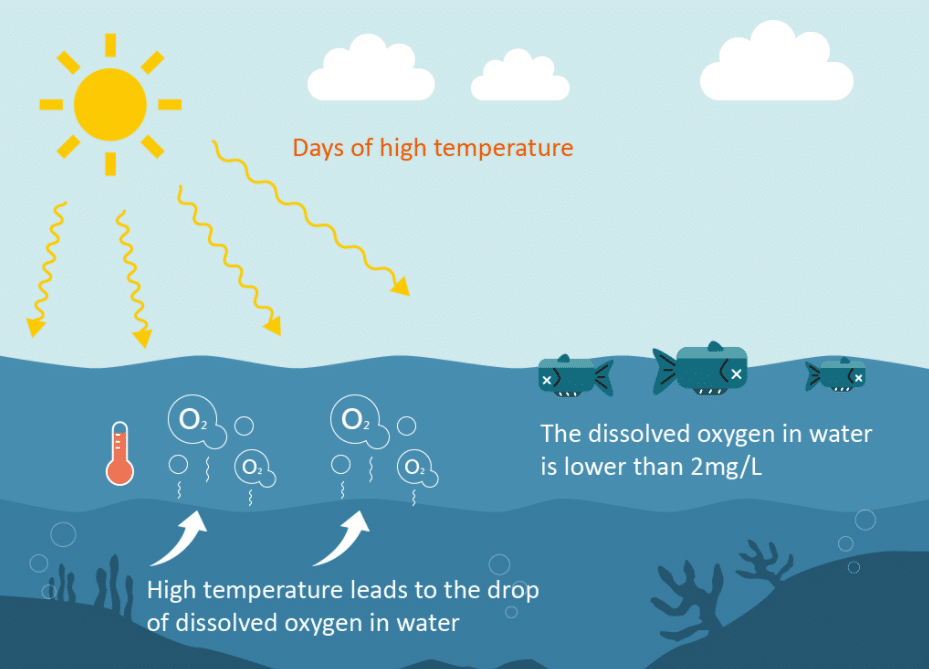
Water temperature is one of the most important factors affecting water systems. Temperature affects dissolved oxygen levels, chemical and biological processes, species composition, water density and stratification, and the life stages of different marine organisms.
For the optimal health of aquatic organisms, temperature must be within its optimum range. Anything outside of this range may adversely affect aquatic organisms; increasing stress levels and often leading to mortality. The reproductive stage of fish (spawning and embryonic development) is the most temperature sensitive period. Temperature also affects ammonia levels in the water, the rate of photosynthesis, the metabolic rate of aquatic organisms, and the sensitivity of aquatic organisms to pollution.
Water temperature fluctuates throughout the day and between seasons due to changes in external environmental conditions. Temperatures in freshwater systems are heated by the sun, and although other water inputs such as precipitation, groundwater, and surface runoff affect water temperature, heat is either lost or gained through condensation and evaporation.
The temperature of the water affects the amount of dissolved oxygen (DO) that the water can hold. As water temperature increases, the amount of dissolved oxygen in the water decreases. DO is the amount of oxygen dissolved in the water, which can also fluctuate daily and seasonally.
DO comes from the atmosphere and photosynthesis of aquatic plants, and is consumed through chemical oxidation and respiration of aquatic organisms (including microorganisms), mainly through the decomposition of organic matter and plant biomass. The optimum pressure of oxygen solubility in water is 1 atm (atmospheric pressure) and ranges from ~15 mg/L at 0ºC to 8 mg/L at 30ºC.
Large fluctuations in DO can disrupt environmental ecosystems affected by changes in runoff, precipitation, and temperature. Fish and other aquatic plants and animals need dissolved oxygen to survive. Some organisms can adapt to changes, however, most cannot. DO also affects the solubility and availability of nutrients in the water.
Conventional variables: pH, total dissolved solids (TDS), conductivity and suspended sediment
Conventional variables are indicators measured to understand the aquatic environment, including watersheds, local environmental conditions, and daily and seasonal variations.
pH (hydrogen potential) is a measure of hydrogen ion concentration ranging from 0 to 14, where 7 is neutral, >7 is basic, and <7 is acidic. Most natural water environments have pH values between 6.0 and 8.5. pH values below 4.5 and above 9.5 are considered lethal to aquatic organisms, while less extreme pH values can interfere with reproduction and other essential biological processes.
Metals, salts and organic compounds are affected by pH. In strongly acidic water, some minerals dissolve in the water, releasing metals and other chemicals. pH may vary depending on different water inputs, such as runoff from land, groundwater, or even drainage from forested areas where weak organic acids and organic matter can change pH.
Total dissolved solids (TDS) concentration is a measure of the dissolved material in a solution. tds includes solutes (sodium, calcium, magnesium, chloride and bicarbonate) that remain as solid residues after the water in the solution/sample has evaporated.
The main sources of TDS are:
- Natural weathering
- Mining
- Industrial waste
- Agriculture
- Sewage
High levels of TDS degrade water quality, making it unsuitable for drinking and irrigation. In general, freshwater TDS levels range from 0 to 1,000 mg/L. This depends on regional geology, climate and weathering processes, as well as other geographic features that affect dissolved oxygen sources and transport to the water system.
Electrical conductivity is a measure of conductive current in µS/cm (micro Siemens/cm). Conductivity in water is influenced by inorganic dissolved solids such as chloride, sulfate, sodium, calcium, etc. The conductivity of streams and rivers is influenced by the geology of the area through which the water flows. In rivers and lakes with outflow, conductivity is typically between 10 and 1,000 µS/cm.
In water, the higher the ion concentration, the more current can be conducted. The conductivity depends on the ionic charge number, the ionic mobility and the temperature.
| Electrical conductivity value | Water type |
| Fresh water | <600 µS/cm |
| Salt | 600-6000 µS/cm |
| Salt water | >6000 µS/cm |
Suspended sediment is the mass of sediment, measured in mg/L, transported by a fluid such as water. Particles are transported by flowing water and settle when the water flow is reduced. Most suspended sediments consist of silt and clay.
During periods of increased water flow, such as rainfall, the concentration of suspended sediment typically increases. Increased levels of suspended sediment reduce light penetration into the water and cause the water to absorb more heat, which raises the water temperature. High concentrations of suspended sediment can move plants, invertebrates and other aquatic organisms that live in the streambed. Increased concentrations can also affect food sources and reduce aquatic fish populations.

Nutrients
Nutrients are essential for the growth and survival of organisms. In addition to other elements such as iron, magnesium and copper, nitrogen and phosphorus are extremely important in aquatic ecosystems.
In aquatic systems, nutrients are present in different chemical forms: organic and inorganic particles, and dissolved organic and dissolved inorganic particles.
During weathering, phosphorus is released from minerals, and some inorganic materials in the soil can bind and prevent phosphorus transport.
Sewage, agricultural fertilizers and animal manure are all artificial sources of nutrients. Elevated nutrient concentrations usually come from direct discharge from wastewater systems or runoff, and excess nitrate increases algal growth, which can lead to eutrophication by limiting primary productivity and promoting the growth of algae (such as blue-green algae).
Eutrophication is a natural process that usually occurs in freshwater ecosystems, however, it can also be an anthropogenic (man-made) process that causes water quality to deteriorate and threatens species survival. As algae (and plants) overgrow, less sunlight penetrates the water, preventing photosynthesis and producing toxins. When plants and algae eventually die and decay, the reduced dissolved oxygen concentration affects aquatic diversity and reduces human use of the water.

Bacteria
E. coli is a type of fecal coliform bacteria from human and animal feces. The Environmental Protection Agency uses E. coli measurements to determine if fresh water is safe for recreational use. Water with elevated E. coli levels may have disease-causing bacteria, viruses and protozoa. Levels of E. coli increase during floods. E. coli is measured by the number of colony-forming units. the EPA’s water quality standard for E. coli is 394 colony-forming units per 100 mL.
Metals
Copper, manganese and zinc are essential for biochemical forms that sustain life, but at high concentrations they can become toxic if ingested by humans and animals, or if consumed by humans exposed to high levels of animals.
Metal toxicity and bioavailability depend on the form and oxidation state in which they occur; dissolved metals are more toxic and bioavailable than metals that are absorbed by sediment or bound to other molecules. Oxidation state, bioavailability, toxicity and solubility are influenced by other water indicators such as pH and dissolved oxygen.
Weathering of rocks and soils, such as erosion and sedimentation, introduces metals into aquatic ecosystems, and the chemical properties of the water will determine how metals are introduced into the sediment. Metals may also occur unnaturally in the water as a result of wastewater treatment, industrial wastes, sewage, contaminated soils, and mining operations.
When metals accumulate in fish, they can be transmitted to humans during consumption. Mercury is particularly susceptible to bioaccumulation and poses a significant risk to human health. The Minamata Bay disaster in Japan in 1968 is a good example. The dumping of industrial waste containing mercury affected thousands of people who consumed local fish and shellfish, which bioaccumulated mercury in their tissues. Many died, some suffered convulsions and paralysis, and pregnant women gave birth to toxic babies with severe deformities such as blindness, deafness, and rough limbs.

Hydrocarbons
Hydrocarbons are organic compounds that contain only carbon and hydrogen.
Polycyclic aromatic hydrocarbons (PAHs) are complex compounds that originate from fossil fuels, organic combustion, and the chemical and biological transformation of organic molecules. They are known to cause cancer and are toxic to aquatic organisms when found in water.
Regulation and control of hydrocarbons in water systems is needed for human health and the safety of aquatic species. Petroleum hydrocarbons are a major pollutant and are often discharged into coastal waters. Bottom sediments are potential hydrocarbon reservoirs that pose a risk to both aquatic animals and humans due to bioaccumulation.

Industrial chemicals
Industrial chemicals can be introduced from industrial waste. Industrial chemicals such as PCBs (polychlorinated biphenyls) threaten aquatic ecosystems and people who regularly consume contaminated fish.
PCBs are known to have negative effects on the immune, neurological, reproductive and endocrine systems of living organisms. PCBs are difficult to break them down in water systems because they are resistant to biological, chemical and thermal degradation.
Ditoxins and furans are toxic organochlorine compounds found in air, water, sediment, animals and food. They come from combustion waste, steel production, and the burning of fossil fuels. When they are present in water, we should be concerned because they are able to accumulate in body fat and bioaccumulate in fish, thus entering the top of the food chain (for humans).

More articles on water quality parameters:
Water Quality Sensors For Water Treatments
What is salinity?
COD VS BOD
3 Main Water Quality Parameters Types
